CORRECTION: Psychedelic Effect Size; Stock Index; DARPA funding; Canadian Circus Update
Hello again,
Sorry to inundate your inbox but I need to make an important correction to today’s post regarding MAPS’ Phase 3 trial.
Michael Haichin pointed out the error in the comments:
“To clarify, MAPS is required to run 2 x 100-person phase 3 trials. Interim analysis confirmed they didn't need to enroll more than 100 people for that first trial (and due to COVID, FDA offered to end early with only 90 subjects). Another phase 3 trial (MAPP2) of 100 people is still needed, so unfortunately there are no savings on time or money.”
In the original I wrote:
“If you recall, MAPS recently announced its Phase 3 trial of MDMA for PTSD has been approved to truncate their enrollment. They were planning to enroll 200 patients but since the interim analysis showed such strong results they are cutting it in half which should save a large chunk of time and money.”
So you can see where I clearly botched it: MAPS’ MDMA Phase 3 trial is made up of two cohorts MAPP1 and MAPP2 each consisting of 100 participants.
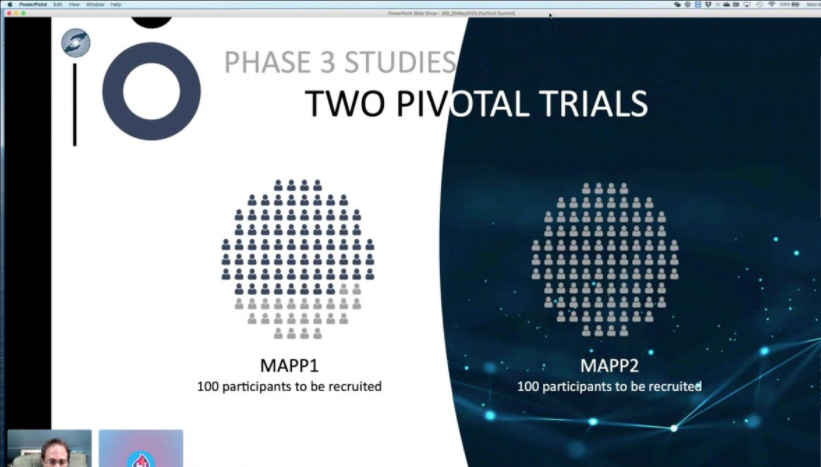
The section from the press release I highlighted pertains only to MAPP1 and the truncated enrollment includes the would-be final 10 participants. MAPS still needs to carry out MAPP2 and will be enrolling participants as COVID restrictions allow.
So the outstanding results do allow MAPS to reduce the total enrollment numbers by 10, not 110.
I regret the error.
Thank you, Michael and MAPS folks for helping correct it.
Thanks,
Zach

