Hello and welcome to The Trip Report Pro dispatch for Friday, June 19th.
Last week ATAI Life Sciences founder Christian Angermayer hosted a fireside chat with CEO Florian Brand and CSO Srinivas Rao.
The full program can be found here and is well worth your time. But if you have Webinar/Zoom fatigue, below is a brief overview of the conversation.
ATAI’s Thesis
The ATAI story is best understood as the scaleable approach to three fundamental trends.
Growing unmet need in mental health, including PTSD, Depression, Anxiety, and Substance Abuse Disorders. Current treatment options for these conditions are failing many, many patients.
Central Nervous System drug discovery and development are historically challenging, which disincentivized investment. However, the science and tools of objective study of Brain and CNS are improving as is the understanding of many mental health conditions.
Scientific and Regulatory Momentum in previously/currently stigmatized substances is becoming less and less of a barrier.
Derisking through compounds that have long-standing recreational/spiritual or previous medical use.
But what does a scaleable application of these insights look like?
A platform, of course.
Platforms are so Hot Right Now
What does it mean to be a platform?
Of course, this question is contingent upon the domain, but in drug development, it seems to be used in two different ways.
Drug discovery “technology platform.” Such a technology ‘platform’ is capable of yielding multiple drug candidates in a specific domain or for certain receptor classes.
Infrastructure platforms that partner/acquire drug programs and offer financial, regulatory, trial development expertise, and guidance. This seems to have been the dominant role of larger pharmaceutical companies that acquire smaller startups to guide regulatory processes and fund many internal research programs.
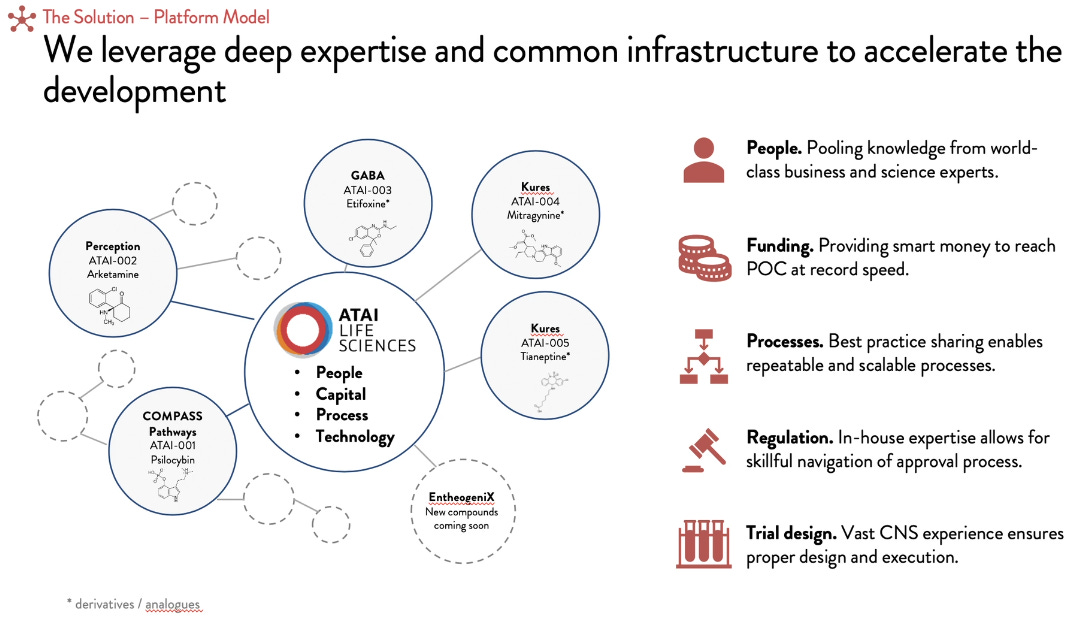
ATAI falls into the second category.
CEO Florian Brand on the ATAI business model:
“I think it’s important to emphasize that we are a platform company… we were one of the first ones to back psychedelic research in a for-profit way with COMPASS. And I think we made a lot of learnings along the way that can be leveraged to make drug discovery and drug development in this field, psychedelic medicine, and more broadly our key indications of anxiety, addiction and depression much more efficient and effective.
That’s when we decided to not only be an investor but let’s pool this knowledge build up this deep domain expertise and make it accessible to the companies that we either start ourselves or acquire and really be the partner of the scientific founders that bring the drug development expertise next to the science to really help to expedite this process in the most responsible way to get these compounds to patients. We believe these economies of scope are essential for fast-paced development.”
It appears that pharma has a long-running on-again, off-again relationship with “platform models.”
This piece was published in Nature’s Bioentreprenuer more than a decade ago advocating for platform models starts with:
“Over the past 20 years, technology-focused platform companies based on partnering business models have come and gone, somewhat cyclically, as large companies and investors bought into their vision and promise only to become disillusioned by their inability to deliver broad solutions or the rapid commodification of the central technology. As a result, customers and investors abandoned platform companies, and those companies in turn abandoned partnering business models in favor of product-focused companies.”
The Roivant Model
I had thought that ATAI’s model most closely resembled a big pharma company that leverages its capital, expertise, and regulatory relations to propel acquisitions of smaller companies into the market. However, just yesterday, I was directed to Roivant Sciences by a reader as the industry model of the ‘startup biotech platform company.’
Roivant, founded in 2014, operates as a platform or holding company that nurtures its 14 subsidiaries developing 28 different compounds.
“The company employs a “hub and spoke” model for research and development, whereby each biopharmaceutical subsidiary functions as an independent entity (“spoke”) using shared resources from the parent (“hub”) and the technology-based subsidiaries focused on different aspects of data science.”
This appears to be the ATAI blueprint.
However, unlike Roivant, whose subsidiaries operate in diverse specialties, ATAI is focused on mental health.
Platform Skepticism
There are two other points of skepticism that I am finding in my research about platform companies.
Platforms as an idea are sexy—the term can be used to elevate valuations in capital markets; if they can land one success, the platform status will increase the likelihood of future achievements. However, such expectations are tough to meet.
On the other hand, allocating capital, expertise, and resources can be diluted across projects. After all, you can only have one favorite child, and portfolio companies will inevitably fall into a pecking order.
The argument for the platform, as the ATAI team puts it, enables scientists and research teams to be who they are and play to their strengths. By providing a “family and emotional support” in addition to financing, regulatory, and trial design expertise allows the founders to do what they do best.
As Angermayer noted:
“These scientists are not necessarily entrepreneurs in the way that Silicon Valley Venture Capital sees entrepreneurs and I don’t believe you should try to make somebody something that they are not but rather build on the strengths that the respective person has.”
Pipeline and Portfolio Companies
Relevant Portfolio projects
COMPASS Pathways: Psilocybin for Treatment-Resistant Depression. The firstborn child in the ATAI family and like many firstborns have the highest expectations. Phase 2 trials are underway, and according to the above graph scheduled for completion in Q1 of 2021 though the impact of COVID I imagine will delay that.
Perception Neuroscience: R-ketamine for Treatment-Resistant Depression. Early research is on identifying a non-psychedelic therapeutic index allowing at-home use.
Kures: Deuterated mitragynine, the active component in Kratom. At lower doses, as an ADHD drug, and at higher doses, it is used to treat pain and intended for opioid use disorder and pain.
Entheogenix: Drug discovery platform looking for novel chemical compounds. Joint venture with Cyclica. The first batch of compounds has already been created and currently working to identify lead candidates.
DemeRx: Ibogaine for Opioid Use Disorder and Opioid Withdrawl. The long-running Ibogaine project headed up by Deborah Mash joined the ATAI family and was announced at the most recent Davos World Economic Summit.
If COMPASS is the firstborn, DemeRx is the favorite.
Here’s Angermayer:
“OK so that last one is one of my favorites because the problem is so big…
Ibogaine will hopefully be a household name like magic mushrooms in a few years. That would make all of us very proud. Ibogaine has the potential to solve the opioid crisis which is a massive problem, mainly in the US… Ibogaine seems to have the potential to erase addiction.”
Florian Brand on Ibogaine:
“It fits in nicely in our sweet spot, it’s derisked in the sense that Ibogaine has been used over the centuries and very recently in clinics to treat opioid use disorder, addiction, to be very precise the extreme cravings when one is getting off these substances and hence increase the success of treating adddiction. So it’s very derisked and meeting a huge unmet need and has a high peak sale potential… The integration component will be critical. Its an even longer trip than psilocybin”
Patent Strategy: Deuterate and Digitalize
Patents and patent strategy in the service of recouping investment and blocking competition may be the most fundamental reason that the pharmaceutical industry is the most vilified in America.
This topic is even more controversial in our field because the psychedelic insight commonly fosters feelings of connection, interdependence, and oneness—traits seemingly at odds with concepts of data exclusivity, market dominance, and blocking access.
This is, of course, compounded by the fact that molecules like LSD, Psilocybin, and MDMA are all in the public domain.
The psychedelic community has taken COMPASS to task on their strategy for psilocybin to date, and the final result remains in the balance.
Rest assured, it is just the beginning.
In regards to how ATAI is thinking about the challenge, Dr. Rao pointed to the various ways of developing and protecting Intellectual Property:
“So we do have composition of matter on several of our companies, the deuterated forms do in fact have composition, they go out quite sometime, certainly into the 2030s. That (Composition) is one of the strongest patents, its what people aspire to and Entheogenix is only looking at new quantities so we’ll have composition there. With other compounds there are other strategies, there are things like CMC (Chemistry Manufacturing and Controls). Sometimes ther are diferent forms of the compound, different crystal or salt forms or the combination of both… that’s something we’re looking at with psilocybin…
The two strategies that caught my attention were:
Deuterating molecules
Digital applications
Deuteration
Deuterium is a non-radioactive isotope of hydrogen that contains one neutron, one proton, and one electron (‘normal’ hydrogen contains one proton and one neutron).
Chemists can link deuterium atoms in place of ‘normal’ hydrogen to carbon atoms in molecules to create significantly different metabolic stability and pharmacokinetics compared to the non-deuterated version.
And to create new compounds in the eyes of the FDA.
In 2017 the FDA decided that deuterated drugs would be considered as New Chemical Entities:
“FDA approved AUSTEDO on April 3, 3017 under NDA 208082 for the treatment of chorea associated with Huntington’s disease. What makes AUSTEDO particularly interesting is that it is the first deuterated drug product approved by FDA. And it is a deuterated version of a previously approved drug: XENAZINE (tetrabenazine) Tablets, which FDA approved on August 15, 2008 under NDA 021894 for the treatment of chorea associated with Huntington’s disease.”
While Deuteration offers a potential avenue for patent protection of previously expired molecules (psilocybin, LSD, MDMA, etc.), there is a tradeoff in that the safety, pharmacokinetics, and duration could be wildly different and thus will need to be explored.
So I wonder how this impacts the efforts and investment to date? Does deuterated psilocybin, for example, need to start from zero? How much of the previous research that points to psilocybin’s safety, efficacy, and pharmacokinetic profile would need to be redone?
(I know someone reading this can answer these questions. I would love to hear your thoughts either by email or in the comments section.)
Digital Strategy
Again Dr. Rao:
“Then the final bit is something we’ve been alluding to… we do actually have a strategy for a combination product with a digital device, a digital therapeutic. That is going to to markedly enhance our ability to block because do in fact have a combination product, we’ve had discussions with multiple regulatory attorneys, you can’t carve out the drug in such a product.”
Digital integration, like patents, be an evergreen topic that we revisit for years and years to come. What does technology-enabled psychedelic therapy look like in 10, 20 years? It’s crazy to think about it. What a time to be alive—simultaneously exciting and terrifying.
But I digress.
Dr. Rao highlighted his hometown of 1,500 in Appalachia as a stark contrast to the San Diego area, where he now lives. In San Diego, it’s relatively easy to find mental health services. In Southeast Kentucky, not so much.
Part of the solution is technology-enabled care that people in less populated areas can access.
How this plays into the patent strategy is above my pay grade but appears to be a cornerstone of the ATAI patent strategy.
De-risking Strategy
Finally, we’ll wrap up with the de-risking strategy.
Christian Angermayer:
“From an investment or a risk point of view the major, major, major advantage of ATAI is that all of the drugs we have talked about have been used in humans extensively, either legally or illegally. So we know a lot about them so the negative surprise risk is dramatically lower than with any other company. Or as I like to say because there are countries where these things are legal, I did extensive due-diligence on our portfolio (cheeky smile).”
Given the history of CNS development and the stubborn nature of PTSD, Depression, Anxiety, Substance abuse, it would appear that such a project is doomed for failure.
But Trip Report readers will be well aware that the hope of psychedelic based treatment is a watershed moment in human history.
The psychedelic experience as spiritual, medicinal, and emotional tools have a long-standing human history across time and culture.
ATAI’s de-risking strategy is best summed up in Ecclesiastes 1:9:
What has been will be again,
what has been done will be done again;
there is nothing new under the sun.
While this may be the case, and most of us would agree, there is still work to be done.
ATAI is arguing that these molecules have been a part of human development for centuries or longer and that to introduce them into the modern healthcare system requires focused expertise in trial design, indication, and endpoint selection.
If you can get that right, the compounds will do the heavy lifting, and the best way to execute on that strategy is to focus exclusively on one area, gather expertise, and iterate and improve upon the process.
ATAI is betting on their particular platform model to best accomplish this goal.
Thank you as always for reading and supporting The Trip Report.
Have a great weekend,
Zach



Another interesting example of deuterium-modification of a drug is work in Bill Fantegrossi's lab led by Michael Berquist that adds deuteriums to the 3,4-methylenedioxy bridge on MDMA, under the hypothesis that it might decrease the ability of enzymes to open the bridge and form toxic metabolites. They show it modestly but significantly alters PK/PD and are currently working on answering the question of whether it alters neurotoxicity.
I would disagree that ibogaine has been derisked by use. Unexpected deaths have occurred in clinical use, while its traditional use has been limited geographically. Other well known traditional natural psychedelics have spread across greater areas and populations. Aside from the known CYP2D6 risk factor, there may be other yet undiscovered risk factors that are rare in the population traditionally using ibogaine.