Software Assisted, Psychedelic Assisted Psychotherapy: part 4/n
The timing of the 'psychedelic renaissance' coincides with another trend in healthcare: Digital Therapeutics

If you are reading this, you are likely up to speed on the progress that MAPS, Usona, and COMPASS Pathways are making towards FDA approval, the many other entrants, and the growing number of policy measures seeking to overturn decades of bad drug policy.
However, there is another emerging trend that is ripening and poised to disrupt healthcare: Digitial Therapeutics (DTx), aka ‘Software as a Drug.’
It’s not even accurate to call it a trend but an established sector.
Rock Health, a leading venture firm in digital health reports, “In the midst of a global pandemic and a US recession, US digital health companies raised $5.4B in venture funding across the first six months of 2020.”
These two emerging trends, psychedelic based therapies, and Digital Therapeutics will inevitably cross-pollinate.
In fact, they already have.
Last week, Field Trip Health released the Trip App.
Previously, ATAI announced Introspect, their Digital Therapeutics platform, and before that, Mindbloom offered virtual ketamine therapy supported by their proprietary software.
The timing of these two emerging trends is not the only coincidence—both are targeting mental health conditions as their first order of business— making the cross-pollination all the more fascinating to watch.
In an episode of the Rock Health Podcast from June 2019 titled Podcast: Navigating pharma-digital therapeutic alliances, Pear Therapeutics CEO Corey M. McCann highlighted the reason why Digital Therapeutics were particularly suited to mental health conditions:
“I think one of the reasons why this is such an interesting field is that we really see this as a whole new therapeutic modality with application on par with things like small and large molecules. We happen to have started in conditions of the brain and specifically speaking different mental health conditions…I think if you’re building a portfolio, you want to ask the question “where will this work? and where is unmet need the highest?” and that’s why you start in CNS. We know that different face-to-face therapeutic paradigms are able to produce a neuroplasticity effect and treat these patients and we know that the pharma pipeline is just about bare for many different mental health conditions.”
The pharmaceutical industry’s recent divestment evidences the challenge of treating mental and behavioral health conditions within the modern healthcare system in the Central Nervous System pipelines:
“In December 2011, Novartis announced that the company is closing its main neuroscience facility in Basel, Switzerland, similarly, GlaxoSmithKline (GSK) and AstraZeneca, both based in the United Kingdom, announced the closure of major parts of their neuroscience research divisions globally. Other pharmaceutical companies such as Merck, Pfizer and Sanofi-Aventis have each closed their research facilities also. This is a major concern for the patients suffering from CNS disorders.”
This trend is an acknowledgment of what the Ancients knew thousands of years ago—you cannot separate the parts from the whole, especially when it comes to the emotions, behavior, psyche, or spirit.
The irony of the tech-as-mental-health-solution is, of course, all the more captivating since the culprit for the rise in particular mental health conditions is tech-enabled platforms like social media.
But unlike social tech unleashed on the masses, DTx seeks to leverage tech for a benefit, and to confirm benefit, these solutions must be rigorously tested.
Pear Therapeutics’ CEO McCann is credited with coining the term ‘Digital Therapeutics’ to denote applications that are meant to be evaluated and scrutinized in the same manner as pharmaceuticals or medical devices. That is through the FDA, with rigorous clinical trials that confirm safety and efficacy, Good Manufacturing Practices (GMP) standards, and are meant to be physician-prescribed and paid for or reimbursed by insurance and third-party payers.
The first Pear products available are for substance use disorder.
Cautious Optimism
A recent spate of articles on the challenges of digital health platforms like Headspace and Talkspace, highlight concerns about efficacy, privacy, and business models at a time when mental health conditions are on the rise and conventional treatments are proving insufficient.
“There is concern that many digital mental health companies are bringing products to market without clinical pilots to demonstrate that their solutions work. Additionally, many of these same companies may claim they are evidence-based when they are based off of vague translations of existing clinical models.”
However, as McCann points out above, this is the difference between digital health and digital therapeutics—FDA approval.
Unfortunately, FDA approval and the establishment of a ‘new branch’ of the Pharmaceutical industry leads many to assume that this new trend will be swallowed up by the greed and bottom-line-before-patients-approach of the most vilified industry in America.
At a time when the mounting costs of healthcare expenditure are blamed on pharmaceutical pricing, digital solutions perhaps offer the possibility of a different form of innovation, one that can make a significant dent on healthcare costs.
In a recent Health Affairs article aptly titled Why Isn’t Innovation Helping Reduce Health Care Costs? The authors make the argument that most innovation in healthcare raises the prices in comparison to the benefits that they deliver.
“National health care expenditures (NHEs) in the United States continue to grow at rates outpacing the broader economy: Inflation- and population-adjusted NHEs have increased 1.6 percent faster than the gross domestic product (GDP) between 1990 and 2018...These patterns exist despite a robust innovation ecosystem in US health care—implying that novel technologies, in isolation, are insufficient to bend the health care cost curve. Indeed, studies have documented that novel technologies directly increase expenditure growth.”
Why doesn’t innovation in healthcare lead to lower prices, higher efficiency, and better outcomes?
Well, the authors argue that there are two ‘flavors’ of healthcare innovation, low-productivity, and high-productivity.
“We believe that the continued expansion of health care costs is largely the result of innovation that tends to have low productivity (exhibit 1). We argue that these archetypes—novel widgets tacked on to existing workflows to reinforce traditional care models—are exactly the wrong properties to reduce NHEs at the systemic level.”
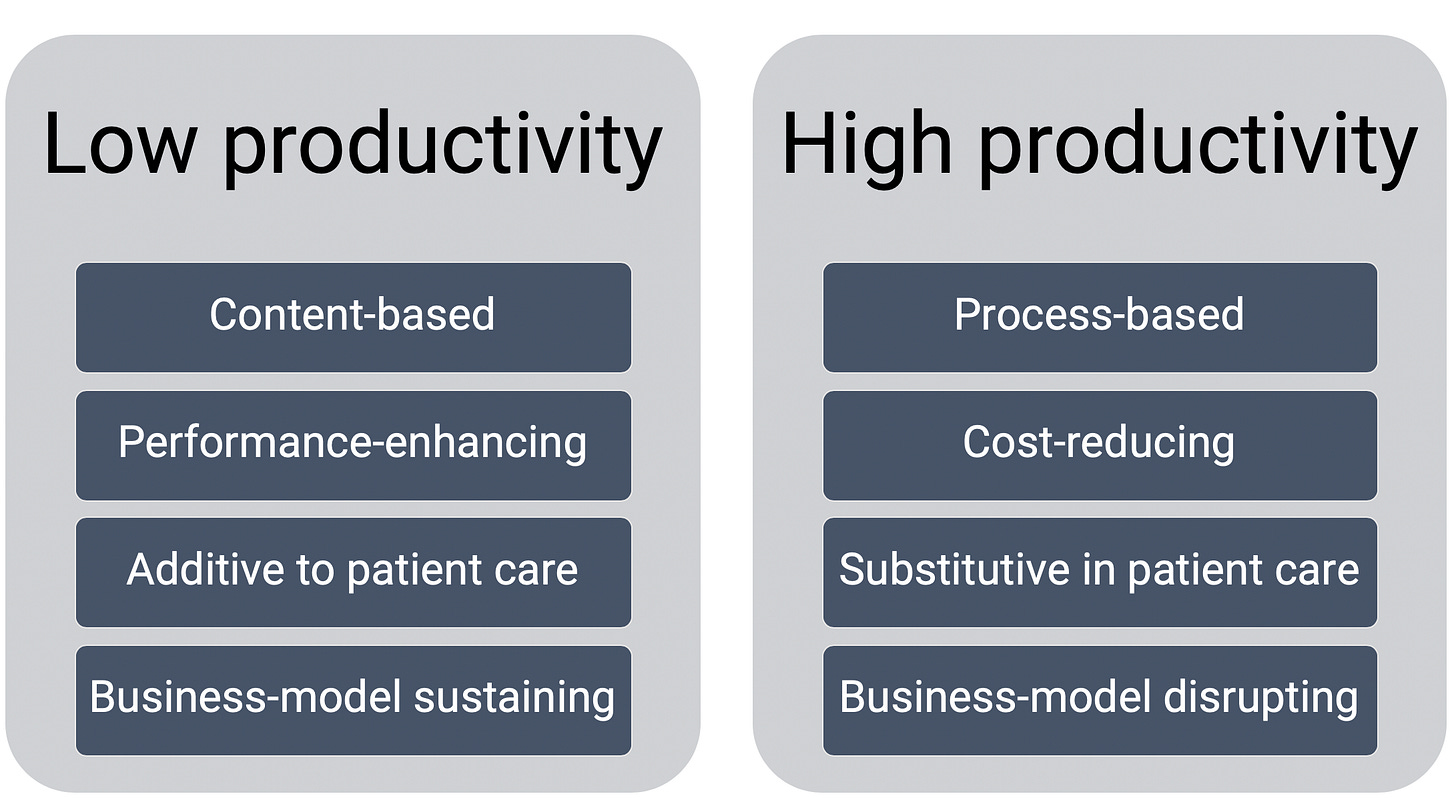
Low productivity innovation includes new biochemical agents (drugs) and imaging technology, which may be better mouse traps but don’t lead to better outcomes necessarily. Low productivity innovation usually offers incremental improvements, but outsized price increases. Low productivity innovation adds layers of complexity to processes such as record keeping, billing, and patient communications—Electronic Medical Records comes to mind.
I am not sure if Healthcare Economists would classify both Psychedelic Assisted Therapy and Digital Therapeutics as High productivity innovation. Still, there is cause for optimism to the extent that they can contribute to more efficient processes, savings, and disruptions to current business models.
For example, facility fees are a scourge of the healthcare system, often adding hundreds or thousands of dollars to a patient’s bill-- how is the rise in telemedicine addressing this?
One promise of Psychedelic Assisted Therapy (PAT) is long term cost savings, by addressing the root of the problem rather than ongoing symptom management a more substantial upfront investment in the more labor-intensive PAT is warranted—would such innovation be considered High productivity? Is this an adequate disruption to current business models?
“The shortcomings in net productivity revealed by the COVID-19 pandemic highlight the urgent need for redesign of health care delivery in this country, and reevaluation of the innovation needed to support it. Specifically, efforts supporting process redesign are critical to promote cost-reducing, substitutive innovations that can inaugurate new and disruptive business models.”
So, is the double-headed serpent of Software Assisted Psychedelic Assisted Psychotherapy a significant high-productivity innovation that could improve outcomes while driving down costs?
I suppose we’ll find out.
News and Headlines
Press Release: Beckley Psytech appoints renowned psychedelic experts to Scientific Advisory Board
DoubleBlind: This Dot-Com Guru is Asking Trump to Legalize Psychedelics
Press Release: Mydecine™ Launches World's First Natural-Sourced cGMP Psilocybin for Global Research, Sales, and Distribution Enterprise
Forbes: Researchers Doubt That Certain Mental Disorders Are Disorders At All
Psilocybin Alpha: Can Taking MDMA with LSD Help Prevent a ‘Bad Trip’? New Clinical Trial Investigates
Lucid News: What Psychedelics Could Mean for Eating Disorders


Upcoming Events
Below are a handful of upcoming events you might find interesting:
MEDIA ALERT: PSFC and MAPS to Provide Major Update on Campaign to Fund Research for MDMA-Assisted Psychotherapy to Treat PTSD



Thanks for reading, see you next time!
Zach

